Oasis Health App Pushes Hype Over Science
Digital apps have reshaped our lives in many important ways. From banking to shopping and even socializing, this technology has enabled a level of convenience and connection previously unheard of. But these apps have a dark side: they're easily misused by people who seek to financially benefit from spreading health misinformation to unsuspecting consumers. One example we encounter frequently on social media is the Oasis Health app.
Oasis presents itself as a “consumer watchdog app," with the company promoting its platform as "revealing the truth behind your food, water, skincare, and everyday products." In practice, however, Oasis makes blatantly false and frequently outrageous claims about safe, regulated products like bottled water--and it charges users for the privilege. The app’s social media videos and ads are full of misleading information, falsely claiming the products are poor quality or unsafe when they are not. These reels do nothing but frighten people, urging them to pay for the Oasis app, which starts at just under $50 per year, while the company's extensive selection of water testing kits cost as much as $636 each.
Let's take a closer look at how the Oasis app functions and why consumers and reporters alike are wrong to rely on its scoring as a reliable metric for determining the safety of bottled water.
Check your sources
The most important fact is that Oasis Health is not a neutral testing authority. It is instead a subjective rating platform that penalizes brands seemingly by whim, but deducting point scores for arbitrary factors such as water type, source and pH levels. Its methodology, a proprietary points-based system, utilizes guidelines based on California’s Public Health Goals (PHGs). The state's Office of Environmental Health Hazard Assessment describes a public health goal as “the level of a chemical contaminant in drinking water that does not pose a significant risk to health. PHGs are not regulatory standards.”
In other words, PHGs bear no resemblance to the Food and Drug Administration’s (FDA) standards of quality (SOQs) for bottled water or the Environmental Protection Agency’s (EPA) public drinking water standards for tap water. As we've noted previously, the FDA is the regulatory body with oversight over bottled water products, protecting consumers of bottled water through the Federal Food, Drug, and Cosmetic Act.
Oasis also relies on test results from the Environmental Working Group, an NGO with a history of promoting fear-based narratives insufficiently rooted in data instead of experts. In 2019, for example, water safety advocates called one of EWG's reports "alarmist and misleading," refusing to call it a "study" because it failed to meet the quality standards scientists are expected to meet in their published work.
Other examples of bias are evident in Oasis' scoring: brands are downgraded for using PET plastic bottles, even though they are the most recycled plastic in the world. Oasis Health routinely exaggerates low-level detections to support its anti-plastic narrative.
The most concerning problem with the Oasis app is most obvious when examining how the media uses it. An article from The Daily Mail (which was recently removed from the internet when we alerted editors to the aforementioned flaws with Oasis) is a typical example: the article’s headline read: "Daily Mail graphic reveals the bottled waters with the most cancer-causing 'forever chemicals." The story claimed widespread chemical contamination in major bottled water brands, relying on testing from Oasis. It alleged “The EPA sets a PFAS exposure limit at 0.4 parts per trillion (ppt), but the health guideline most researchers agree on is 0.1 ppt.”
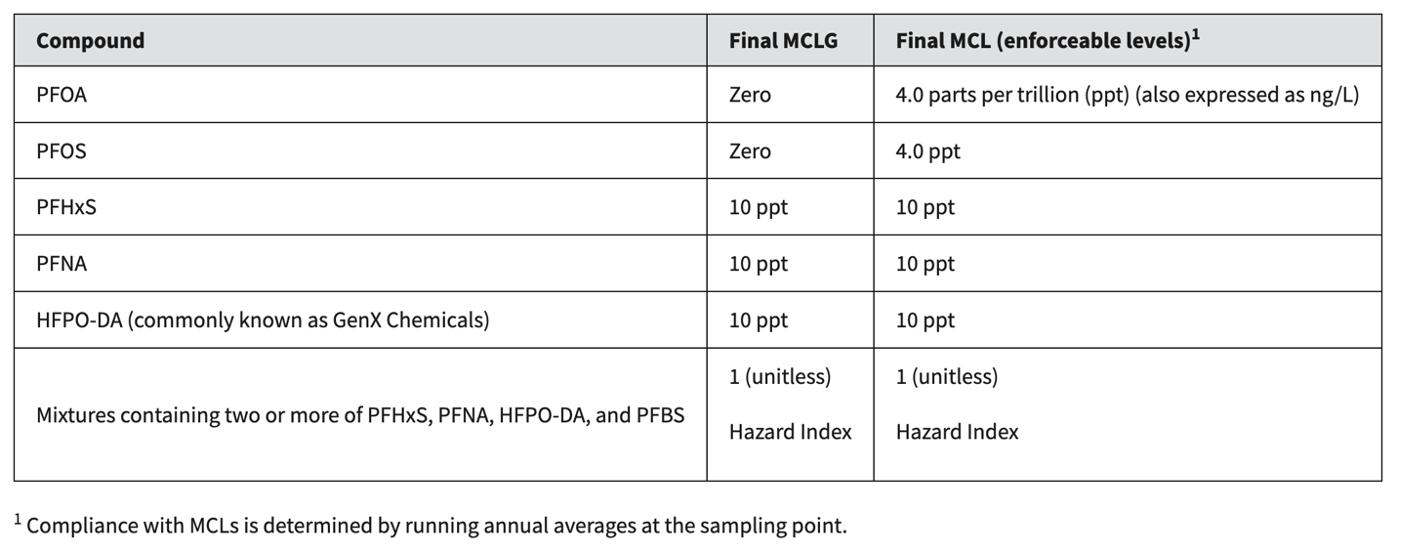
The problem? This is false. By citing figures that are 10 (or more) times lower than the EPA's PFAS levels, Oasis falsely claims that the list of brands exceeded levels or guidelines. Information about the EPA’s PFAS standards is available online, which should be easy for the media to verify, but in this case, they did not. In reality, the detected levels of PFAS chemicals in the bottled water brands mentioned in this story were all well below EPA limits, which are in the chart below.
In addition, the FDA completed a PFAS detection analysis of 197 domestic and imported bottled water samples collected at retail locations across the U.S. between 2023 and 2024. The 197 samples included purified, artesian, spring, and mineral waters. Ten of the samples analyzed by the FDA had detectable levels of PFAS. However, none of those samples had PFAS levels that exceeded the EPA’s maximum contaminant levels (MCLs) for PFAS in tap water. They also did not exceed the International Bottled Water Association’s (IBWA) PFAS SOQs.
Currently, the FDA does not have an SOQ for PFAS in bottled water (or any other food products). However, since 2019, IBWA members have been required to test their bottled water products annually for PFAS and comply with an SOQ. That same year, IBWA asked FDA to establish an SOQ for PFAS in bottled water. The FDA responded to IBWA’s request by stating that “establishing an SOQ for PFAS in bottled water at this time would not significantly enhance FDA’s mission of public health protection.”
Ultimately, Oasis Health's testing guidelines appear designed to frighten consumers away from bottled water—aligning with environmental activism rather than providing objective risk assessment. This is fearmongering taking the place of critical scientific analysis. Relying on advocacy-driven apps rather than regulatory guidelines gives consumers a distorted view of product safety. Bottled water remains a safe, convenient option for healthy hydration.